Describe Thomson's Concept of the Atom.
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Thomson in the late 19th century.

Thomson Model Of The Atom Ppt Video Online Download
Thomsons discovery of the electron completely changed the way people viewed atoms.
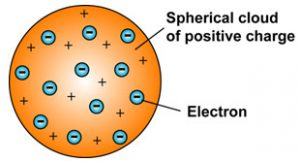
. Although Thomsons model explained that atoms are electrically neutral the results of experiments carried out by other scientists could not be explained by this model as we will see below. He also said that negative and positive charges are equal in magnitude so an atom as a whole is electrically neutral. At the time of discovery JJ.
Neutron was not considered in Thomsons model of the atom. Describe thomsons concept of the atom. The Plum Pudding Model is a model of atomic structure proposed by JJ.
Plum pudding is an English dessert similar to a blueberry muffin. JJ Thomson Proposed that an atom consists of a positively charged sphere and the electron was embedded into it. In which he describe the resin as electrons and spherical body as central body in which electrons.
The current knowledge of atoms and atomic theory has been informed by many scientists going back to Aristotle and Democritus. Prior to Thomsons discovery the atom was thought to be indivisible. He believed that the electrons were like dry fruits in a.
Describe Thomsons concept of the atom. The electrons were assumed to be positioned in revolving circles around the atom in this model to be having a cloud of positive charge. Up until the end of the 19th century atoms were thought to be tiny solid spheres.
The positive charge is. Thomson called this negatively charged particle a corpuscles. He stated that atoms consist of a positively charged sphere and electrons are embedded into it.
In 1903 Thomson proposed a model of the atom consisting of positive and negative charges present in equal amounts so that an atom would be electrically neutral. Now at least part of the atom was to be composed of Thomsons particulate negative corpuscles although the rest of the positively charged part of the atom remained somewhat nebulous and ill-defined. Though several alternative models were advanced in the 1900s by Kelvin and others Thomson.
Thomson was the first to put forward a model to explain the structure of an atom. Thomsons atomic model is also called water melon model or Christmas pudding model. In magnitude the negative and the positive charges were equal.
Thomsons atomic model William Thomson also known as Lord Kelvin imagined the atom as a sphere with a positive charge evenly distributed and embedded in it enough electrons to neutralize the positive charge. Thomsons Plum Pudding Model is the first model to represent the atomic structure of matter. Following the discovery of the electron JJ.
The positive and negative charge is equal in magnitude and therefore an atom has no charge as a whole and is electrically neutral. Physics 21062019 1930 314180. Thomson developed what became known as the plum pudding model in 1904.
Precisely speaking an atom consists of three major subatomic particles which are protons neutrons and electrons. Soon after the discovery of the electron the atomic model was proposed saying that the structure of an atom is like a plum pudding. Learn about the contributions made to early atomic theory by.
Thomson atomic model earliest theoretical description of the inner structure of atoms proposed about 1900 by William Thomson Lord Kelvin and strongly supported by Sir Joseph John Thomson who had discovered 1897 the electron a negatively charged part of every atom. With this model Thomson abandoned his earlier nebular atom hypothesis in which the atom was composed of immaterial vortices. Thomson Atomic Theory.
He compared the electrons with the raisins in the spherical Christmas pudding and to seeds in a watermelon. Thomson had discovered that atoms are composite objects made of pieces with positive and negative charge and that the negatively charged electrons within the atom were very small compared to the entire atom. Thomson model of atom is the structure of an atom proposed by the scientist JJThomson who was the first person to discover the electron.
2 Get Other questions on the subject. In Thomsons model the atom is composed of electrons surrounded by a soup of positive charge to balance the electrons negative charges like negatively charged plums surrounded by positively charged pudding. Jj thomson proposed his model of atom in the year 1903according to him-An atom consists of a sphere of positive charge with negatively charged electrons embeded in itThe positive and negative.
According to Thomsons model the atom is like a plum pudding structurally. Describe Thomsons concept of the atom. In Thomsons plum pudding model of the atom the electrons were embedded in a uniform sphere of positive charge like blueberries stuck into a muffin.
According to Thomsons Plum Pudding Model a substance is consists of small spheres which are having the radius of about 10-10 m in diameter. Thomsons concept of the atom was referred to as the plum pudding model because it was randomly distributed electrons throughout an atom. According to the postulates of Thomsons atomic model an atom resembles a sphere of positive charge with electrons negatively charged particles present inside the sphere.
JJ Thomsons Model of Atom Thomson proposed a theory according to which he defined atoms to be similar to that of a Christmas pudding. Thomson model of atom is described base on three main facts.
J J Thomson The History Of The Atom

A Describe Thomson S Model Of The Atom Which Subatomic Particle Was Not Present In Thomson S Model Of The Atom Science Shaalaa Com
Comments
Post a Comment